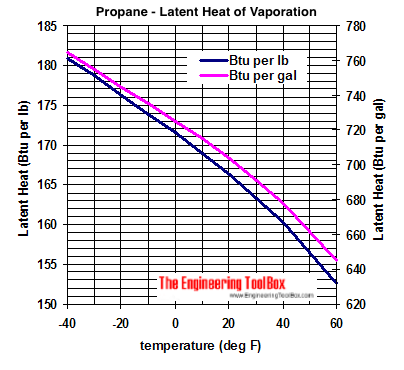
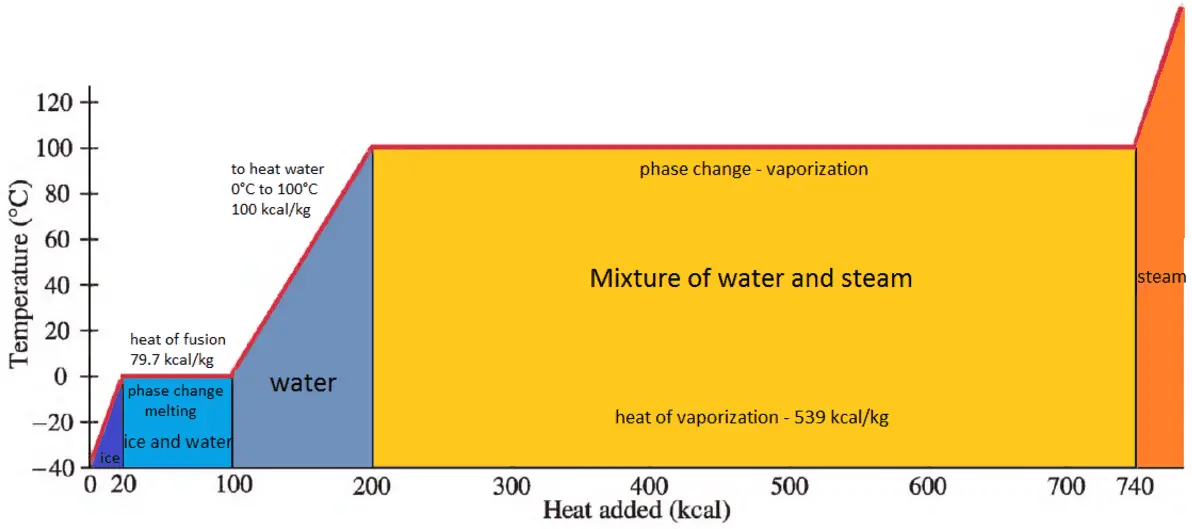
SOLVED:Calculate the heat change at 100^∘ C for each of the following and indicate whether heat was absorbed or released: a. calories to vaporize 10.0 g of water b. joules to vaporize
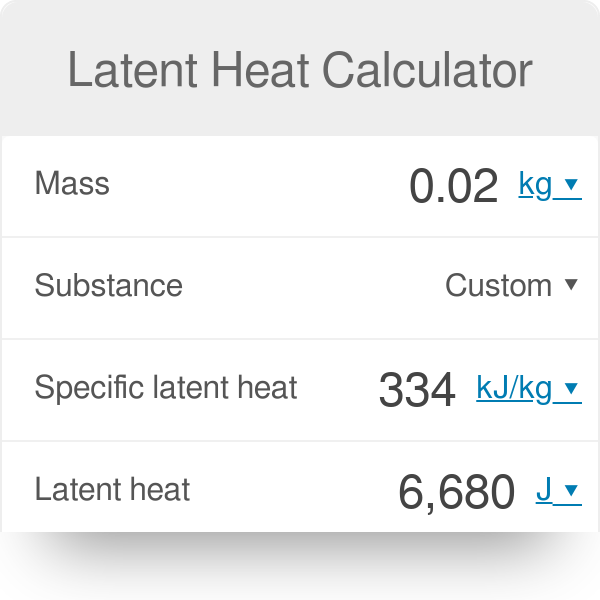
A swimmer coming out from a pool is covered with 18 g water film. How much heat must be supplied to evaporate the water at 298 K? What is the internal energy
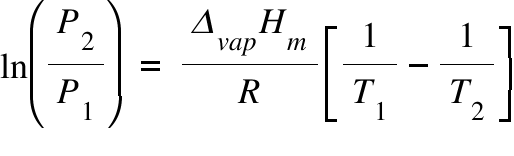
Correlation of Vapor Pressure at Different Temperatures by Clausius Clapeyron Equation Calculator | Calistry

Latent heat of vaporization of nanofluids: Measurements and molecular dynamics simulations: Journal of Applied Physics: Vol 118, No 1

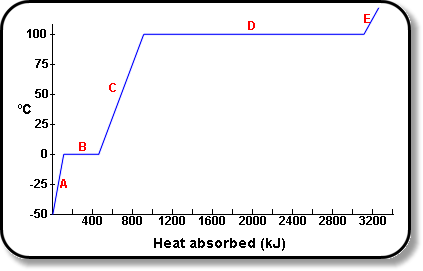
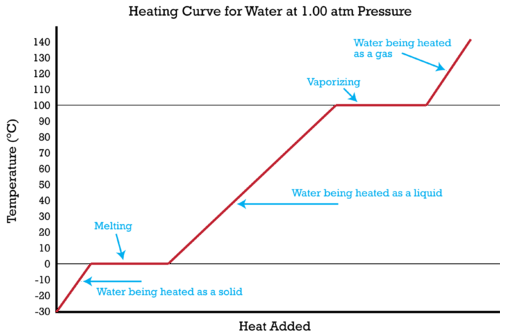



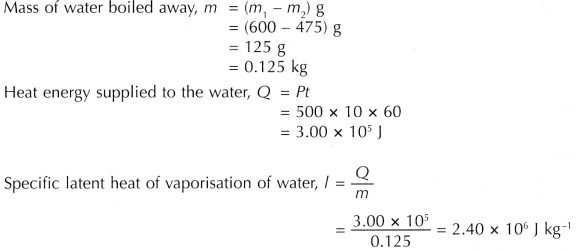



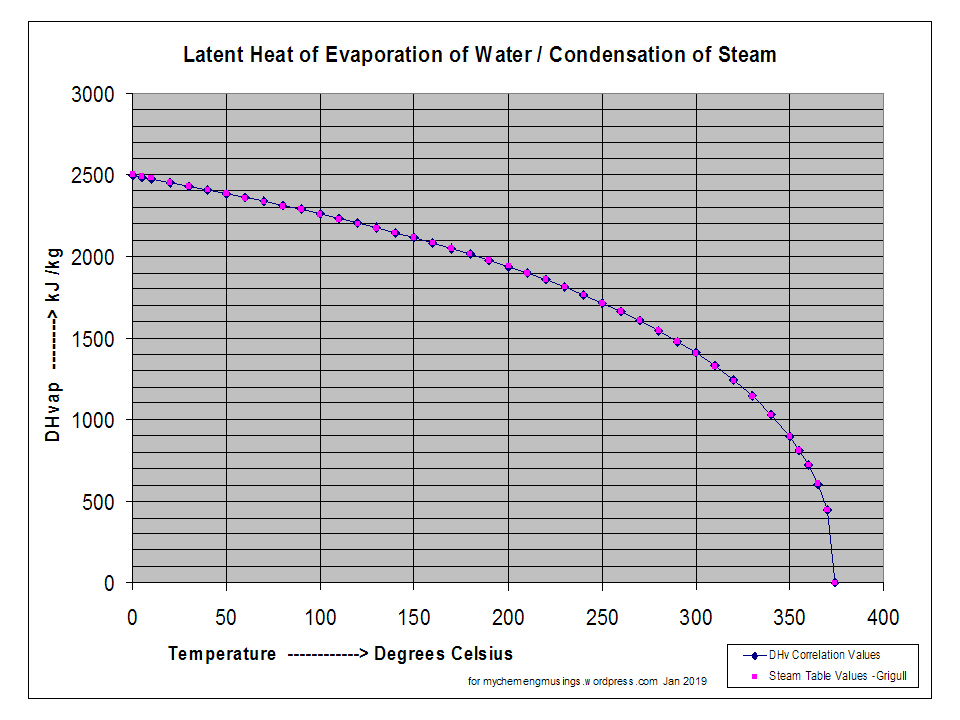



![Latent heat of vaporization for main components of LNG [10]. | Download Table Latent heat of vaporization for main components of LNG [10]. | Download Table](https://www.researchgate.net/publication/330572654/figure/tbl3/AS:718422421803010@1548296661881/Latent-heat-of-vaporization-for-main-components-of-LNG-10.png)