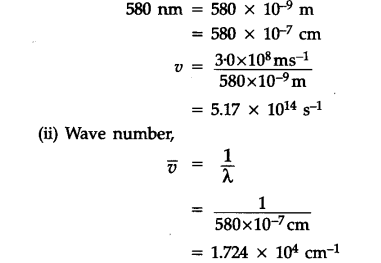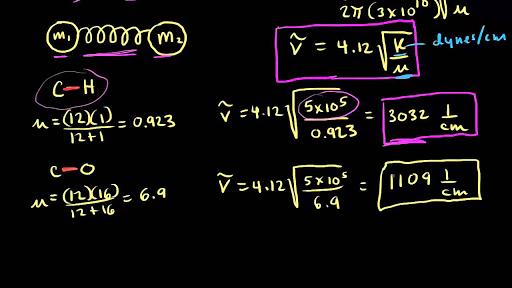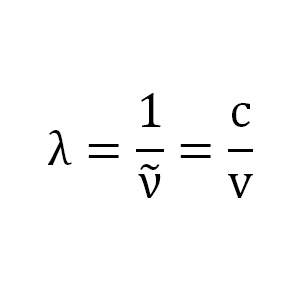Calculate the wave number for the shortest wavelength transition in the Balmer series of atomic hydrogen
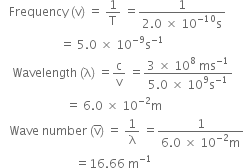
Calculate the wavelength, frequency and wave number of light wave whose period is 2.0 x 10-10 s. - Zigya

Calculate the wave number for the longest wavelength transition in the Balmer series fo atomic hydrogen . ( R(H) = 109677 cm^(-1)).

calculate the wave number and wavelength of the first two lines in a paskin series of hydrogen spectrum 2tynlvxx -Chemistry - TopperLearning.com